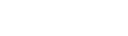Excimer lamps/Excimer irradiation Units Outstanding productivity with the world's highest illuminance
and highest exposure
Since becoming the world’s first supplier in 1993, we have been supporting many customers as a top manufacturer of excimer lamps.
We offer a broad range of support: from economical test equipment to cutting-edge process development and production facilities.
- Features
- Example
- Options
- FAQs
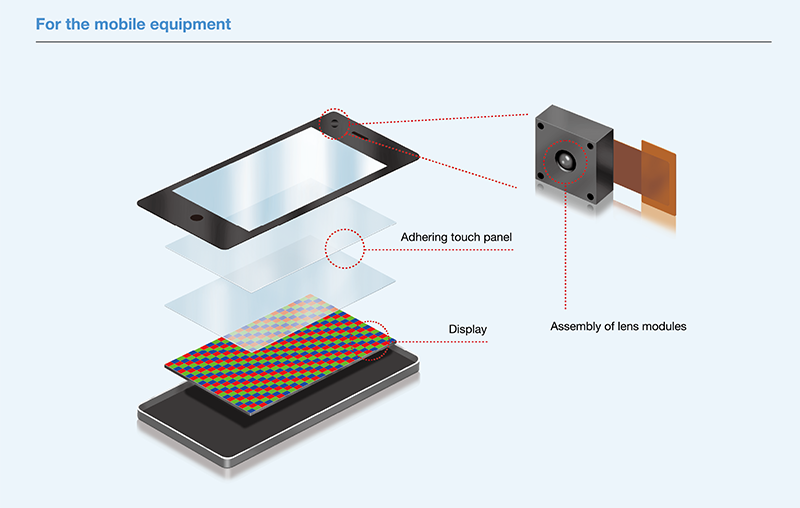
- Excimer Irradiation Units for surface treatment
- 1.Suitable for various applications:
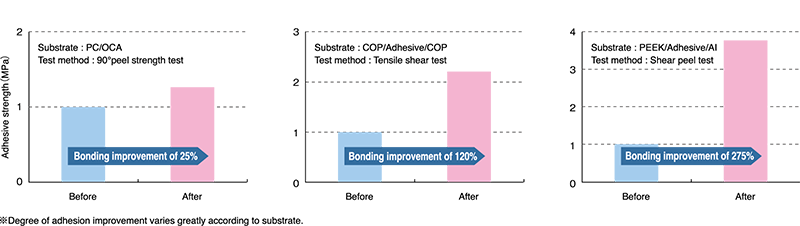
Dry cleaning, improving hydrophilicity (wettability), improving adhesion, ashing/residue removal, etc. - 2.High photon energy (7.2 eV):
Can quickly generate highly reactive oxygen atoms(O3p、O1D) and various radicals - 3.Low-temperature processing:
Because no mercury is used, lamps run cool and emit few wavelengths that increase temperature - 4.Instantaneous ignition:
CoO is improved by lighting only when required for processing - 5.Diverse line-up:
Achieves the industry's highest illuminance, highest exposure, and longest-life lamps.
From small products to long lengths, from test equipment to automatic transport production facilities
-
Heating

-
Ozone filter unit

-
Oxygen concentration monitor

-
Environmental ozone sensor

-
Irradiance meter

-
Is light-shielding eyewear needed?
Yes, we recommend wearing light-shielding eyewear.
Light at 172 nm is rapidly attenuated in air, but longer-wavelength light is also emitted at about 1/100th of the intensity at 172 nm.
While there is little effect at low illuminance, at high illuminance or for long-term operation,
light-shielding eyewear is advisable. -
Are N2 utilities required?
There are models for which N2 is essential, models in which it improves efficiency, and models for which it is unnecessary.
Advise us of your usage environment/conditions, processing speed, and etc.
if you would like us to propose the most suitable system. -
How should exhaust of ozone be handled?
Common handling methods include drafters and local exhausting of the processing space through ducts.
Purge gas in the lamp-housing/process space also needs to be exhausted.
Regarding safety-related specifications, please observe the installation precautions and contact our sales representative for details. -
Do you offer irradiation experiment/demonstration systems for loan?
Yes we do. Irradiation experiments and evaluations are performed at our laboratory in Himeji City, Hyogo Prefecture.
We have analytical systems such as SEM, XPS, and FT-IR that enable surface analyses at lower cost than evaluation institutions.
Another advantage is that we can analyze a surface immediately after irradiation, avoiding issues due to surface changes that can easily occur after irradiation. -
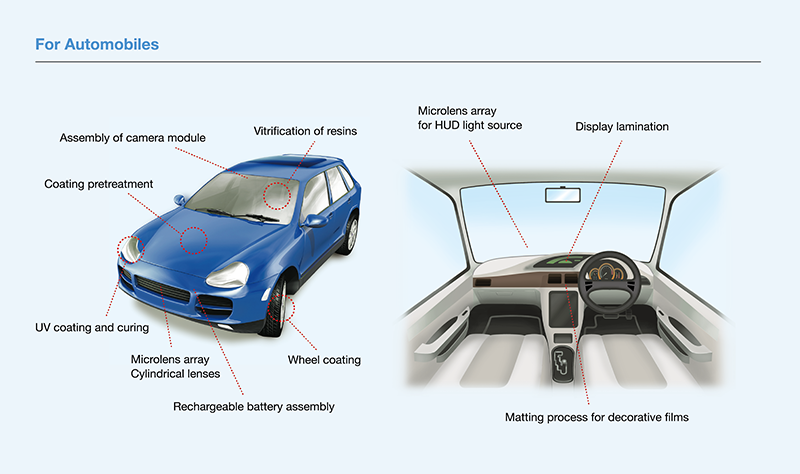
Can the cleaning effect be obtained in air?
It is possible to obtain a cleaning effect even in air.
However, if the distance from the lamp to the irradiated object is large, the light will be attenuated and the effect will be reduced.
Be careful when setting the irradiation distance when conducting irradiation experiments (recommended: about 2 to 4 mm)
By controlling the atmosphere, it is possible to extend the irradiation distance and irradiate three-dimensional objects. -
What is the optimal atmosphere and irradiation distance?
Approximately 2 to 4 mm is recommended for atmospheric air conditions.
Optimal parameters must be determined when changing the oxygen concentration in the processing atmosphere or extending the irradiation distance.
For reference, here are the parameters for changing the contact angle of the glass substrate.
(Vertical axis: contact angle after irradiation; Horizontal axis: oxygen concentration at the time of irradiation)
Catalog Download
Requests and Inquiries
Inquiries and requests for information
Inquiries by telephone 03-5657-1033